Provide personalised and coordinated care for patients suffering from advanced chronic diseases through personalised healthcare plans. These goal seeks to improve quality of life, early detection and evaluation of the deterioration, recovery or the delay of the deterioration, and the empowerment of patients and caregivers.
ESPECIFIC OBJETIVES:
- Provide collaboration tools for care teams to create personalised healthcare plans addressing the multi-dimensional nature of the patients.
- Implement smart tools supporting clinical decision-making
- Provide a more active role for patients and caregivers in the management of their own health, promoting shared decision-making and individualised adaptive interventions.
- Achieve improvements in the health of the patient, delaying their deterioration, enhancing symptom control, and avoiding exacerbation and adverse events.
- Improve patients’ autonomy, satisfaction and quality of life.
- Achieve higher efficiency of integrated care: use of resources, coordination between all agents, and professional working conditions.
- Deploy the intervention in seven different European and associated regions.
ADLIFE is divided into 11 Work Packages (WP), four transversal (TRANS) and seven technical packages (TS) :
- WP1 “Coordination and Management” (TRANS)
- WP2 “Dissemination and communication” (TRANS)
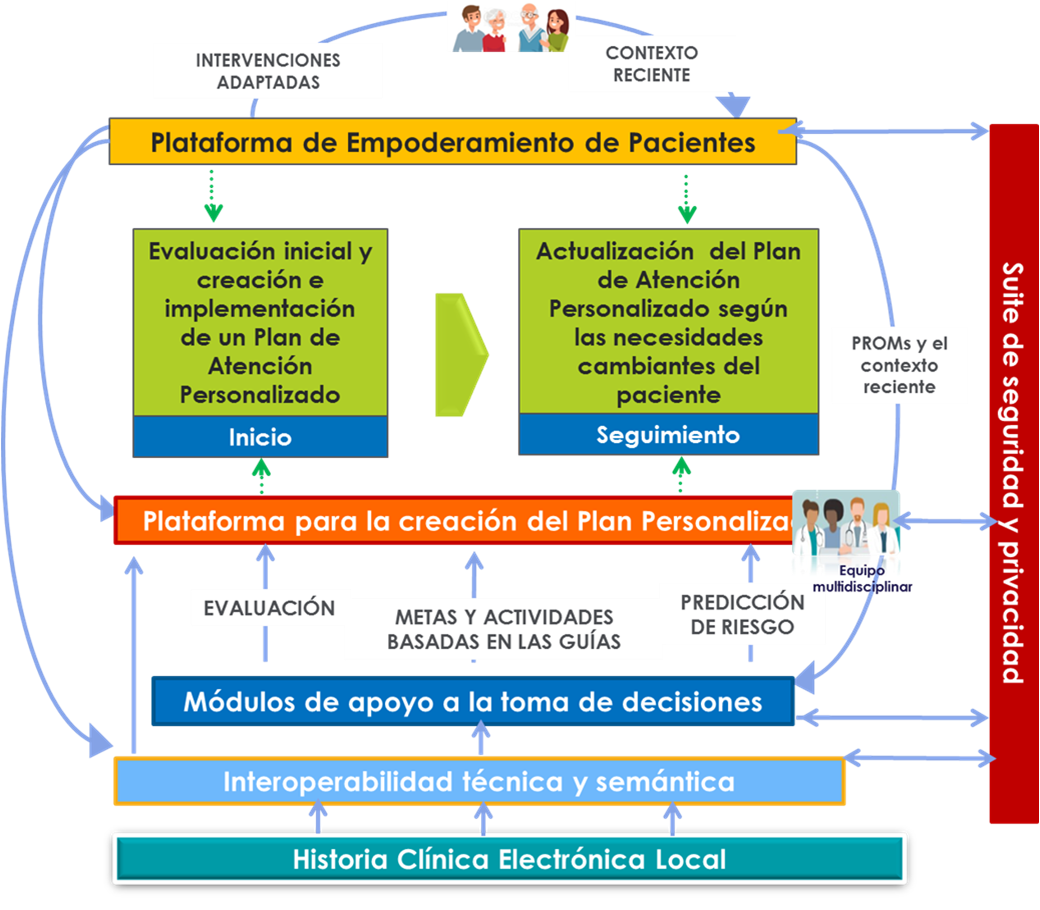
- WP3 “Digital tools to create a personalised healthcare plan management platform” (TS)
- WP4 “Patient empowerment platform and personalised mechanisms for self-care support” (TS)
- WP5 “Support systems for clinical decision-making and early alert tools” (TS)
- WP6 “Personalised evidence-based care” (TS)
- WP7 “Empowerment of patients, caregivers, and communities” (TS)
- WP8 “Pilot design and implementation” (TS)
- WP9 “Evaluation” (TS)
- WP10 “Exploitation” (TRANS)
- WP11 “Ethical Requirements” (TRANS)
ADLIFE will be deployed through pilot projects in seven countries and healthcare systems including the Basque Country (Osakidetza), United Kingdom (NHSL Lanarkshire), Denmark (Southern Denmark), Poland (FALKHOSP Lower Silesia), Germany (Werra-Meißner Kreis), RJH-Sweden (Region Jämtland Härjedalen) and Israel (Maccabi Healthcare Services).
The intervention involves 577 health professionals from 75 hospitals, clinics, and primary care centres, and 882 patients and 1243 caregivers.
- Implement an innovative solution that will facilitate the customisation of the healthcare plans and improve their capacity for a prompt response and early detection of the needs and preferences of the patients.
- Offer support services for clinical decision-making, which in addition to proposing goals and treatment, anticipate their patients’ changing needs and recommend specific services for early action against preventable events.
- Facilitate the provision of specific services that allow for higher patient participation levels in the management of their own health.
- Demonstrate that the ADLIFE solution improves patient health and healthcare results, contributing to the continuity of coordinated care.
Kronikgune, as the project coordinator, is in charge of the coordination and management of the project (WP1). It also leads work package 9, thus being responsible for assessing the pilot programmes and analysing their results. Kronikgune is also responsible for Task 7.1 “Results reported by the patient and surveys to measure the health of patients and their quality of life in relation to health and well-being”. Kronikgune will also participate in the pilot study in the Basque Country supporting Osakidetza in different tasks such as the definition of the personalised healthcare plan management, the incorporation of the Patient Empowerment Platform (PEP) app in the Basque Country, and the design and development of the clinical decision support system.
- Coordination: Lola Verdoy, Ana Ortega-Gil, Bárbara López, Irati Erreguerena, Jose Maria Aguirre, Janire Flores, Ane Fullaondo, Esteban de Manuel
- Main team: Laura de la Higuera Vila, Urko Aguirre Larrakoetxea, Jose María Quintana López, Beñat Zubeltzu Sese, Remedios Vega Iñigo.
- Evaluation: Ania Gorostiza, Borja García Lorenzo, Nerea González Hernández, Igor Larrañaga, Javier Mar Medina.
List of partners of the consortium:
- University of Strathclyde (United Kingdom)
- University of Warwick (United Kingdom)
- Odense Universitetshospital (Denmark)
- Szpital Specjalistyczny im. A. Falkiewicza we Wrocławiu (Poland)
- OptiMedis AG (Germany)
- Region Jämtland Härjedalen (RJH) (Sweden)
- Assuta Ashdod ltd (Israel)
- SRDC (Turkey)
- everis (Spain)
- The European Institute for Innovation through Health Data (I-HD)(Belgium)
- Kronikgune Institute for Health Services Research(Spain)
Organisations within the Osakidetza:
- Alto Deba IHO
- Araba IHO
- Barrualde-Galdakao IHO
- Bidasoa IHO
- Bilbao-Basurto IHO
- Debabarrena IHO
- Donostialdea IHO
- Ezkerraldea-Enkarterri-Cruces IHO
- Tolosaldea IHO
- Uribe IHO
IHO = Integrated Health Organization

 The life expectancy of people with chronic diseases has grown significantly in numerous countries due to the advances in medical sciences.
The life expectancy of people with chronic diseases has grown significantly in numerous countries due to the advances in medical sciences.